4ivt
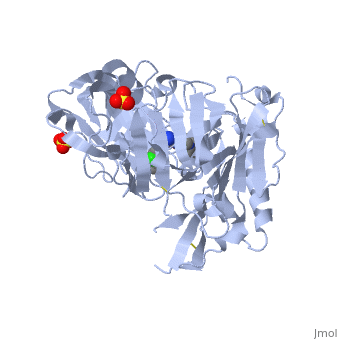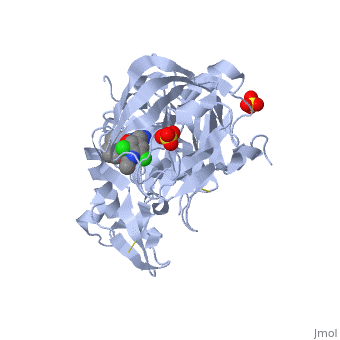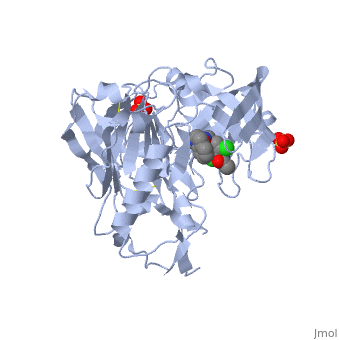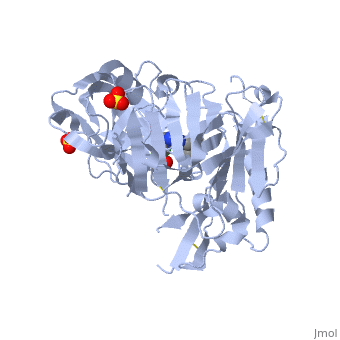
Crystal structure of BACE1 with its inhibitorCrystal structure of BACE1 with its inhibitor
Structural highlights
FunctionBACE1_HUMAN Responsible for the proteolytic processing of the amyloid precursor protein (APP). Cleaves at the N-terminus of the A-beta peptide sequence, between residues 671 and 672 of APP, leads to the generation and extracellular release of beta-cleaved soluble APP, and a corresponding cell-associated C-terminal fragment which is later released by gamma-secretase.[1] [2] Publication Abstract from PubMedProteolytic cleavage of amyloid precursor protein by beta-secretase (BACE1) is a key step in generating the N-terminal of beta-amyloid (Abeta), which further forms into amyloid plaques that are considered as the hallmark of Alzheimer's disease. Inhibitors of BACE1 can reduce the levels of Abeta and thus have a therapeutic potential for treating the disease. We report here the identification of a series of small molecules bearing an indole acylguanidine core structure as potent BACE1 inhibitors. The initial weak fragment was discovered by virtual screening, and followed with a hit-to-lead optimization. With the aid of co-crystal structures of two discovered inhibitors (compounds 19 and 25) with BACE1, we explored the SAR around the indole and aryl groups, and obtained several BACE1 inhibitors about 1,000-fold more potent than the initial fragment hit. Accompanying the lead optimization, a previously under-explored sub-site opposite the flap loop was redefined as a potential binding site for later BACE1 inhibitor design. Virtual screening and structure-based discovery of indole acylguanidines as potent beta-secretase (BACE1) inhibitors.,Zou Y, Li L, Chen W, Chen T, Ma L, Wang X, Xiong B, Xu Y, Shen J Molecules. 2013 May 16;18(5):5706-22. doi: 10.3390/molecules18055706. PMID:23681056[3] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences
|
| ||||||||||||||||||