2f4m: Difference between revisions
No edit summary |
No edit summary |
||
| (10 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
==The Mouse PNGase-HR23 Complex Reveals a Complete Remodulation of the Protein-Protein Interface Compared to its Yeast Orthologs== | |||
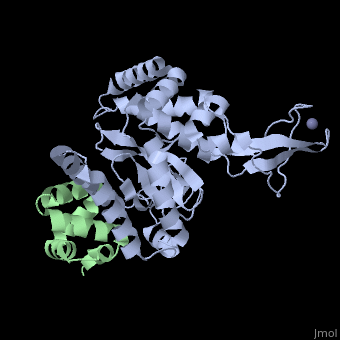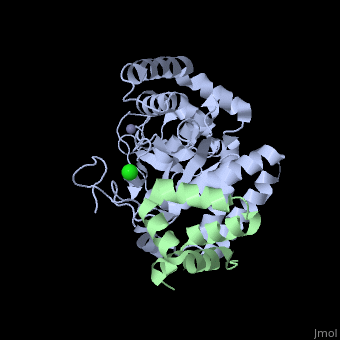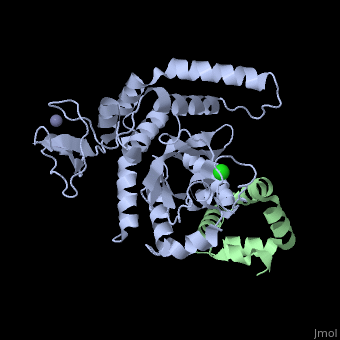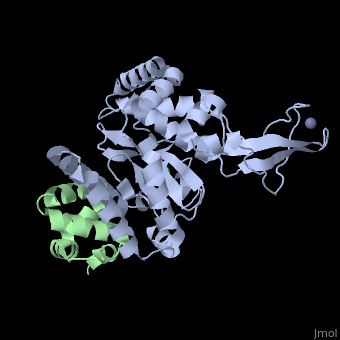
<StructureSection load='2f4m' size='340' side='right'caption='[[2f4m]], [[Resolution|resolution]] 1.85Å' scene=''> | |||
| | == Structural highlights == | ||
<table><tr><td colspan='2'>[[2f4m]] is a 2 chain structure with sequence from [https://en.wikipedia.org/wiki/Mus_musculus Mus musculus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2F4M OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2F4M FirstGlance]. <br> | |||
</td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 1.85Å</td></tr> | |||
<tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> | |||
<tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2f4m FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2f4m OCA], [https://pdbe.org/2f4m PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2f4m RCSB], [https://www.ebi.ac.uk/pdbsum/2f4m PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2f4m ProSAT]</span></td></tr> | |||
</table> | |||
== Function == | |||
[https://www.uniprot.org/uniprot/NGLY1_MOUSE NGLY1_MOUSE] Specifically deglycosylates the denatured form of N-linked glycoproteins in the cytoplasm and assists their proteasome-mediated degradation. Cleaves the beta-aspartyl-glucosamine (GlcNAc) of the glycan and the amide side chain of Asn, converting Asn to Asp. Prefers proteins containing high-mannose over those bearing complex type oligosaccharides. Can recognize misfolded proteins in the endoplasmic reticulum that are exported to the cytosol to be destroyed and deglycosylate them, while it has no activity toward native proteins. Deglycosylation is a prerequisite for subsequent proteasome-mediated degradation of some, but not all, misfolded glycoproteins.<ref>PMID:11562482</ref> <ref>PMID:12606569</ref> <ref>PMID:15358861</ref> | |||
== Evolutionary Conservation == | |||
[[Image:Consurf_key_small.gif|200px|right]] | |||
Check<jmol> | |||
<jmolCheckbox> | |||
<scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/f4/2f4m_consurf.spt"</scriptWhenChecked> | |||
<scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview03.spt</scriptWhenUnchecked> | |||
<text>to colour the structure by Evolutionary Conservation</text> | |||
</jmolCheckbox> | |||
</jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=2f4m ConSurf]. | |||
<div style="clear:both"></div> | |||
<div style="background-color:#fffaf0;"> | |||
== Publication Abstract from PubMed == | |||
Peptide N-glycanase removes N-linked oligosaccharides from misfolded glycoproteins as part of the endoplasmic reticulum-associated degradation pathway. This process involves the formation of a tight complex of peptide N-glycanase with Rad23 in yeast and the orthologous HR23 proteins in mammals. In addition to its function in endoplasmic reticulum-associated degradation, HR23 is also involved in DNA repair, where it plays an important role in damage recognition in complex with the xeroderma pigmentosum group C protein. To characterize the dual role of HR23, we have determined the high resolution crystal structure of the mouse peptide N-glycanase catalytic core in complex with the xeroderma pigmentosum group C binding domain from HR23B. Peptide N-glycanase features a large cleft between its catalytic cysteine protease core and zinc binding domain. Opposite the zinc binding domain is the HR23B-interacting region, and surprisingly, the complex interface is fundamentally different from the orthologous yeast peptide N-glycanase-Rad23 complex. Different regions on both proteins are involved in complex formation, revealing an amazing degree of divergence in the interaction between two highly homologous proteins. Furthermore, the mouse peptide N-glycanase-HR23B complex mimics the interaction between xeroderma pigmentosum group C and HR23B, thereby providing a first structural model of how the two proteins interact within the nucleotide excision repair cascade in higher eukaryotes. The different interaction interfaces of the xeroderma pigmentosum group C binding domains in yeast and mammals suggest a co-evolution of the endoplasmic reticulum-associated degradation and DNA repair pathways. | |||
Structure of the mouse peptide N-glycanase-HR23 complex suggests co-evolution of the endoplasmic reticulum-associated degradation and DNA repair pathways.,Zhao G, Zhou X, Wang L, Li G, Kisker C, Lennarz WJ, Schindelin H J Biol Chem. 2006 May 12;281(19):13751-61. Epub 2006 Feb 24. PMID:16500903<ref>PMID:16500903</ref> | |||
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |||
</div> | |||
<div class="pdbe-citations 2f4m" style="background-color:#fffaf0;"></div> | |||
== | ==See Also== | ||
Peptide N-glycanase | *[[Peptide N-glycanase|Peptide N-glycanase]] | ||
== References == | |||
== | <references/> | ||
__TOC__ | |||
</StructureSection> | |||
[[Category: Large Structures]] | |||
[[Category: Mus musculus]] | [[Category: Mus musculus]] | ||
[[Category: Kisker C]] | |||
[[Category: Lennarz WJ]] | |||
[[Category: Kisker | [[Category: Schindelin H]] | ||
[[Category: Lennarz | [[Category: Wang L]] | ||
[[Category: Schindelin | [[Category: Zhao G]] | ||
[[Category: Wang | [[Category: Zhou X]] | ||
[[Category: Zhao | |||
[[Category: Zhou | |||
Latest revision as of 08:12, 17 October 2024
The Mouse PNGase-HR23 Complex Reveals a Complete Remodulation of the Protein-Protein Interface Compared to its Yeast OrthologsThe Mouse PNGase-HR23 Complex Reveals a Complete Remodulation of the Protein-Protein Interface Compared to its Yeast Orthologs
Structural highlights
FunctionNGLY1_MOUSE Specifically deglycosylates the denatured form of N-linked glycoproteins in the cytoplasm and assists their proteasome-mediated degradation. Cleaves the beta-aspartyl-glucosamine (GlcNAc) of the glycan and the amide side chain of Asn, converting Asn to Asp. Prefers proteins containing high-mannose over those bearing complex type oligosaccharides. Can recognize misfolded proteins in the endoplasmic reticulum that are exported to the cytosol to be destroyed and deglycosylate them, while it has no activity toward native proteins. Deglycosylation is a prerequisite for subsequent proteasome-mediated degradation of some, but not all, misfolded glycoproteins.[1] [2] [3] Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. Publication Abstract from PubMedPeptide N-glycanase removes N-linked oligosaccharides from misfolded glycoproteins as part of the endoplasmic reticulum-associated degradation pathway. This process involves the formation of a tight complex of peptide N-glycanase with Rad23 in yeast and the orthologous HR23 proteins in mammals. In addition to its function in endoplasmic reticulum-associated degradation, HR23 is also involved in DNA repair, where it plays an important role in damage recognition in complex with the xeroderma pigmentosum group C protein. To characterize the dual role of HR23, we have determined the high resolution crystal structure of the mouse peptide N-glycanase catalytic core in complex with the xeroderma pigmentosum group C binding domain from HR23B. Peptide N-glycanase features a large cleft between its catalytic cysteine protease core and zinc binding domain. Opposite the zinc binding domain is the HR23B-interacting region, and surprisingly, the complex interface is fundamentally different from the orthologous yeast peptide N-glycanase-Rad23 complex. Different regions on both proteins are involved in complex formation, revealing an amazing degree of divergence in the interaction between two highly homologous proteins. Furthermore, the mouse peptide N-glycanase-HR23B complex mimics the interaction between xeroderma pigmentosum group C and HR23B, thereby providing a first structural model of how the two proteins interact within the nucleotide excision repair cascade in higher eukaryotes. The different interaction interfaces of the xeroderma pigmentosum group C binding domains in yeast and mammals suggest a co-evolution of the endoplasmic reticulum-associated degradation and DNA repair pathways. Structure of the mouse peptide N-glycanase-HR23 complex suggests co-evolution of the endoplasmic reticulum-associated degradation and DNA repair pathways.,Zhao G, Zhou X, Wang L, Li G, Kisker C, Lennarz WJ, Schindelin H J Biol Chem. 2006 May 12;281(19):13751-61. Epub 2006 Feb 24. PMID:16500903[4] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences
|
| ||||||||||||||||||