InhA
3D structures of ACP family
InhAby Kelly Hrywkiw IntroductionThe enzyme InhA is coded from the INHA gene that is similar in sequence to the Salmonella typhimurium gene which plays a role in fatty acid synthesis, and is part of a short chain dehydrogenase/reductase family[1][2]. Inha is an NADH dependent trans enoyl-acyl ACP carrier protein that is part of the fatty acid biosynthesis system: fatty acid synthase two (FASII), and plays a role in the synthesis of Mycolic Acid[3][2]. Mycolic acids are long chain fatty acids (C54 to C63) that are essential in cell wall formation of the human pathogen Mycobacterium tuberculosisas well as other mycobateria such as Mycobacterium leprae, and are associated with virulence[4]. InhA has been proposed as the target of the thioamide drugs, ethionamide (ETH) and protionamide (PTH), which have been used in treatment of mycobacterial infections [2]. However stains of M. tuberculosis that are resistant to thioamide drugs have been increaseing worldwide, and therefor research into the exact mechanisms of these drugs is of importance.
Structure of InhA
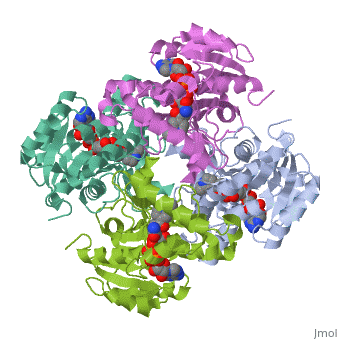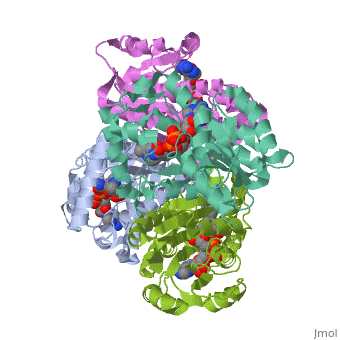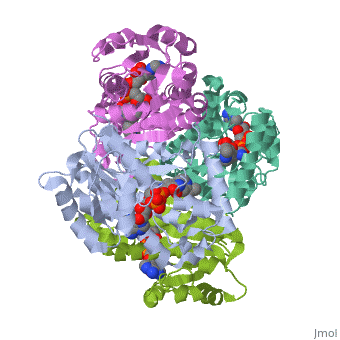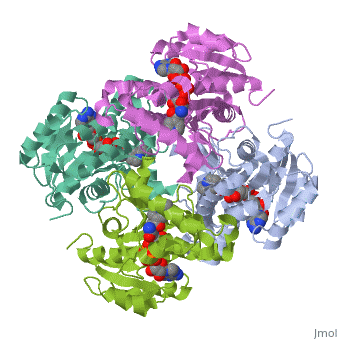
 The InhA enzyme of M. tuberculosis is a homotetramer (Fig. 1) composed of a repeating subunit of a single domain with a Rossmann Fold in the core that provides a NADH binding site[5]. The single domain can be broken down into two substructures that are connected by short peptide loop[1][5]. The overall structure exhibits α/β folding with a series of α helices flanking a central β sheet of multiple parallel β strands[5]. Substructure 1 of InhAconsists of 6 parallel β strands and 4 α helices interwoven together to form a core α/β structure that contains the n-terminal domain[1]. The first substructure can be further broken down into two sections, the consisting of two β strands and two short α helices [1]. The first section is connected to the by a β strand that crosses over the two domains, and leads into the second section initiating at the third α helix [1]. (A-3) is connected by a long loop to a 14 residue β strand that then leads into the fourth α helix [1]. A-4 then leads into a fifth strand β , followed by a 25 residue α helix , and into the final strand β [1]. Substructure 2 of InhAcontains the c-terminal region of the molecule and consists of a small β strand , and two α helices which are connected by a short five residue loop[1]. The C-terminal domain consits of two other α helices [1]. Hydrophobic Binding PocketInhA contains a where ligands bind to a higly conserved binding site[3]. The site is lined with the hydrophobic residues tyrosine 158 (Y158), phenylalanine 149 (F149) methionine 199 (M199), trypotophan 222 (W222), leucine 218 (K218), methionine 161 (M161), and proline 193 (P193)[3]. The fatty acyl binding site is also located in the hydrophobic pocket of InhA and consists primairly of the substrate binding loop [6].
InhA's Function in the Mycolic Acid Pathway InhA plays a key role in the synthesis of fatty acids, particularly in M. tuberculosis which, has type one fatty acid synthesis (FASI) and type two fatty acid synthesis (FASII) which together function in the synthesis of mycolic acids[8]. FASI synthesizes C16-18 and C24-26 fatty acids. The fatty acids from FASI are then sent to FASII which promotes chain extension, forming long-chain meromycolic acids that are 56-64 carbons in length[9]. The final step in FASII is completed by InhA which reduces 2-trans-enoyl-ACP's with chain lengths over twelve carbons in a NADP dependent manner where the hydride transfer precedes protonation(Fig. 2)[8][10]. The reaction takes place as follows: initially NADH binds to the active site mediated by van der Waal interactions with the side chains of phenylalanine 41 (F41), leucine 218 and methionine 155 to the phosphate group of NADH. There are additional interaction with lysine 165 that also mediate binding[10][5]. Binding of NADH causes a conformational change in the Aspartate 42 and Arginine 43 side chains and an over all conformational change in InhA[5][3]. In addition tyrosine 158 plays an important role in aligning the carbonyl substrate, in fact; rotation about its Cα-Cβ bond by 60° brings it into a position where it can hydrogen bond to the carbonyl of the 2-trans enoyl-ACP and provide it with electrophilic stabilization[10]. The substrate binds in a U-shaped conformation with its trans double bond adjacent to the nicotinamide ring of NADH[6]. Inha then reduces the 2-trans double bond of the substrate by forming a enoyl intermediate through the transfer of a hydride ion from NADH to the third carbon of the substrate, followed by protonation of the second carbon[5]. The binding of both the substrate and the cofactor induces another conformational change in InhA that allows for the release of the meromycolic acid product[5]. The meromycolic acids undergo claisen condensation with a C26 fatty acid followed by reduction to a mature mycolic acid[9][5].
InhA and Thioamide Drugs The primary target of the thioamide drugs PTH and ETH have been shown to be InhA in both genetic and molecular experiments[3]. Both PTH and ETH require activation by various cellular componets to form the NAD adduct that acts to inhibit InhA, and therefore connot be studied in in vitro[3]. The exacct mechanism of their activation is still under speculation, however a flavin monooxygenase (EthA) has been shown to participate in ETH and PTH activation[3]. In fact, strains of M. tuberculosis that have mutations in the gene which express EthA exhibit resistance to thioamide drugs[3]. Currently studies are being carried out to determine other methods of treatment for mycobaterial infections that dont require activation by cellular constituents, due to the incerease of drug resistant cases world wide. The ETH-NAD adduct and the PTH-NAD adduct (P1H) have been found to occupy the same hydrophobic pocket of InhA as NADH and exhibit the same van der Waal interactions between and the ethyl or proply group with distances of 3.3Å and 3.2Å respectively[3]. EAD or P1H binding forces the rotation of by 90° which causes a ring stacking interation with the pyridine ring on the adduct. In addtion π stacking interactions form between the propyl group of P1H and the ethyl group of at distance of ~3.3Å. These interations and conformational changes in InhA contribute to its inactivation. Developmentaly this is important, for InhA is no longer active an the mycolic acids nessasary in cell wall compostion of various mycobacteria will not be formed.
Protein SuperfamilyInhA can be categorized into two distinct families, SDR's and ACP's. The SDR FamilyInhA can also be classified into a family of short chain dehydrogenase/reductases (SDR's). This family consists of proteins exhibiting a central core with a Rossmann fold that contains a NADH binding site. There are approximately 3000 primary structures outlines in various sequence databases[11]. Examples of proteins in this family are listed below with links to their corresponding proteopedia page.
The ACP familyInhA can be further classified into the acyl carrier protein family(ACP's). These proteins generally all function in the transport of substrates in a myriad of pathways, such as: the synthesis of polypeptides and fatty acids[12]. |
| ||||||||||
Additional ResourcesAdditional Resources
- Mycobacterium tuberculosis InhA bound with ETH-NAD adduct, in the RCSB Protein Data Bank
- Mycobacterium tuberculosis InhA bound with PTH-NAD adduct, in the RCSB Protein Data Bank
- Crystal structure of wild-type InhA:NADH complex, in the RCSB Protein Data Bank
ReferencesReferences
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Sacchettini, James (New Rochelle, NY) 1999 INHA crystals and three dimensional structure United States Albert Einstein College of Medicine of Yeshiva University (Bronx, NY) 5882878 http://www.freepatentsonline.com/5882878.html
- ↑ 2.0 2.1 2.2 Molle V, Gulten G, Vilcheze C, Veyron-Churlet R, Zanella-Cleon I, Sacchettini JC, Jacobs WR Jr, Kremer L. Phosphorylation of InhA inhibits mycolic acid biosynthesis and growth of Mycobacterium tuberculosis. Mol Microbiol. 2010 Dec;78(6):1591-605. doi:, 10.1111/j.1365-2958.2010.07446.x. Epub 2010 Nov 9. PMID:21143326 doi:10.1111/j.1365-2958.2010.07446.x
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Wang F, Langley R, Gulten G, Dover LG, Besra GS, Jacobs WR Jr, Sacchettini JC. Mechanism of thioamide drug action against tuberculosis and leprosy. J Exp Med. 2007 Jan 22;204(1):73-8. Epub 2007 Jan 16. PMID:17227913 doi:10.1084/jem.20062100
- ↑ Jmol - a paradigm shift in crystallographic visualization. J. Appl. Cryst. (2010). 43, 1250-1260 doi:https://dx.doi.org/10.1107/S0021889810030256
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Dias MV, Vasconcelos IB, Prado AM, Fadel V, Basso LA, de Azevedo WF Jr, Santos DS. Crystallographic studies on the binding of isonicotinyl-NAD adduct to wild-type and isoniazid resistant 2-trans-enoyl-ACP (CoA) reductase from Mycobacterium tuberculosis. J Struct Biol. 2007 Sep;159(3):369-80. Epub 2007 May 3. PMID:17588773 doi:http://dx.doi.org/10.1016/j.jsb.2007.04.009
- ↑ 6.0 6.1 Rozwarski DA, Vilcheze C, Sugantino M, Bittman R, Sacchettini JC. Crystal structure of the Mycobacterium tuberculosis enoyl-ACP reductase, InhA, in complex with NAD+ and a C16 fatty acyl substrate. J Biol Chem. 1999 May 28;274(22):15582-9. PMID:10336454
- ↑ Wilson M, DeRisi J, Kristensen HH, Imboden P, Rane S, Brown PO, Schoolnik GK. Exploring drug-induced alterations in gene expression in Mycobacterium tuberculosis by microarray hybridization. Proc Natl Acad Sci U S A. 1999 Oct 26;96(22):12833-8. PMID:10536008
- ↑ 8.0 8.1 Gurvitz A, Hiltunen JK, Kastaniotis AJ. Function of heterologous Mycobacterium tuberculosis InhA, a type 2 fatty acid synthase enzyme involved in extending C20 fatty acids to C60-to-C90 mycolic acids, during de novo lipoic acid synthesis in Saccharomyces cerevisiae. Appl Environ Microbiol. 2008 Aug;74(16):5078-85. Epub 2008 Jun 13. PMID:18552191 doi:10.1128/AEM.00655-08
- ↑ 9.0 9.1 Bhatt A, Brown AK, Singh A, Minnikin DE, Besra GS. Loss of a mycobacterial gene encoding a reductase leads to an altered cell wall containing beta-oxo-mycolic acid analogs and accumulation of ketones. Chem Biol. 2008 Sep 22;15(9):930-9. PMID:18804030 doi:10.1016/j.chembiol.2008.07.007
- ↑ 10.0 10.1 10.2 Parikh S, Moynihan DP, Xiao G, Tonge PJ. Roles of tyrosine 158 and lysine 165 in the catalytic mechanism of InhA, the enoyl-ACP reductase from Mycobacterium tuberculosis. Biochemistry. 1999 Oct 12;38(41):13623-34. PMID:10521269
- ↑ Oppermann U, Filling C, Hult M, Shafqat N, Wu X, Lindh M, Shafqat J, Nordling E, Kallberg Y, Persson B, Jornvall H. Short-chain dehydrogenases/reductases (SDR): the 2002 update. Chem Biol Interact. 2003 Feb 1;143-144:247-53. PMID:12604210
- ↑ Rafi S, Novichenok P, Kolappan S, Zhang X, Stratton CF, Rawat R, Kisker C, Simmerling C, Tonge PJ. Structure of acyl carrier protein bound to FabI, the FASII enoyl reductase from Escherichia coli. J Biol Chem. 2006 Dec 22;281(51):39285-93. Epub 2006 Sep 29. PMID:17012233 doi:10.1074/jbc.M608758200