1fno
PEPTIDASE T (TRIPEPTIDASE)PEPTIDASE T (TRIPEPTIDASE)
Structural highlights
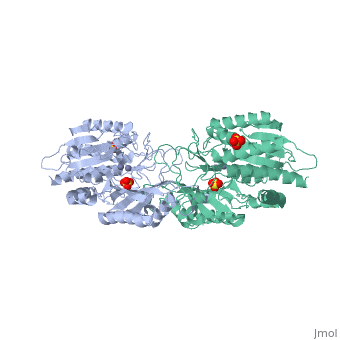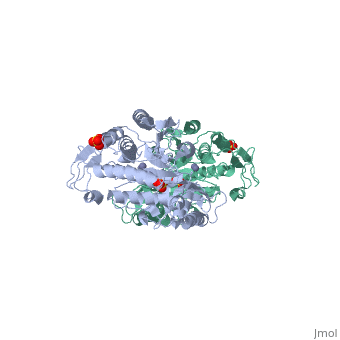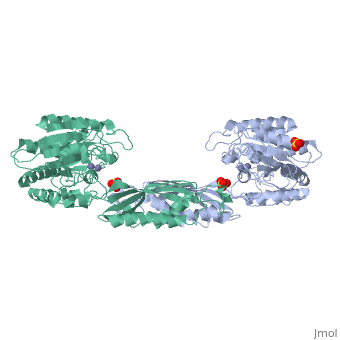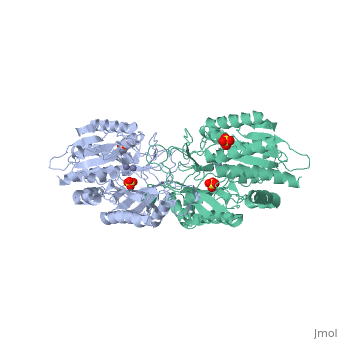
Function[PEPT_SALTY] Cleaves the N-terminal amino acid of tripeptides. Hydrolyzes tripeptides containing N-terminal methionine, leucine, or phenylalanine. Displays little or no activity against dipeptides, N-blocked or C-blocked tripeptides, and tetrapeptides.[1] Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. Publication Abstract from PubMedThe structure of peptidase T, or tripeptidase, was determined by multiple wavelength anomalous dispersion (MAD) methodology and refined to 2.4 A resolution. Peptidase T comprises two domains; a catalytic domain with an active site containing two metal ions, and a smaller domain formed through a long insertion into the catalytic domain. The two metal ions, presumably zinc, are separated by 3.3 A, and are coordinated by five carboxylate and histidine ligands. The molecular surface of the active site is negatively charged. Peptidase T has the same basic fold as carboxypeptidase G2. When the structures of the two enzymes are superimposed, a number of homologous residues, not evident from the sequence alone, could be identified. Comparison of the active sites of peptidase T, carboxypeptidase G2, Aeromonas proteolytica aminopeptidase, carboxypeptidase A and leucine aminopeptidase reveals a common structural framework with interesting similarities and differences in the active sites and in the zinc coordination. A putative binding site for the C-terminal end of the tripeptide substrate was found at a peptidase T specific fingerprint sequence motif. Structure of peptidase T from Salmonella typhimurium.,Hakansson K, Miller CG Eur J Biochem. 2002 Jan;269(2):443-50. PMID:11856302[2] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences
|
| ||||||||||||||||||