Student Projects for UMass Chemistry 423 Spring 2016
Understanding the chemical basis of disease and life processes
InstructionsInstructions
under construction...
HelpHelp
Questions & AnswersQuestions & Answers
Here are some from me and previous students. Post new ones at Sandbox 423.
- For step-by-step instructions on creating example scenes, try Proteopedia:DIY:Scenes.
- A very useful color scheme is "chain" which colors separate proteins or DNA strands in different colors (first select all protein or DNA).
- To show the biological unit, follow directions at Biological Unit: Showing. The pdb file will display the "asymmetric unit" = the smallest unit that can be replicated to generate the full crystal. Example: the protein may function as a dimer (you need biochemical experiments to tell you this -- crystallography and NMR won't tell you), but the pdb file may display a monomer (if the dimer is symmetric) or two dimers (if they have slightly different conformations in the crystals -- perhaps due to crystal contacts or perhaps representing 2 functional states of the protein!).
- Instructions for references are at Help:Editing#Citing_Literature_References.You can follow the format used in the example on the Asp receptor and they will be put in automatically.
You just find out the PMID code (listed in pubmed for example) and insert it into the following, at the place where you want the reference cited (click edit to see what is actually inserted here). [1] You also need to add the section:
References
- ↑ Yeh JI, Biemann HP, Pandit J, Koshland DE, Kim SH. The three-dimensional structure of the ligand-binding domain of a wild-type bacterial chemotaxis receptor. Structural comparison to the cross-linked mutant forms and conformational changes upon ligand binding. J Biol Chem. 1993 May 5;268(13):9787-92. PMID:8486661
- Hey guys this is just a useful tip:
If you get an xml error after you try to save your changes it is due to the green scene coding. Our group experienced this issue and it would not let us access our sandbox. In order to fix this go back (or find the page to edit in your history) and delete the green scene code that was just entered. Then save the page and you should be back to your sandbox. This may be trivial to many, but just throwing it out there.
- To highlight some interesting portion of your protein:
Under the selections tab, you can "limit to residue numbers." So for example enter in 60-65, then click "replace selection" below. Then if you go to the colors tab you can pick a color for just the residues you have selected. If it is a loop or if they are hard to see you can go to the representation tab and set selection to ball and stick or spacefill.
It is also useful to click the "selection halos:" box under the picture. That shows you what you have in your selection.
- If you suddenly can't get to your sandbox page (error message XML error: Mismatched tag at line 1), try Help:Errors
Tips from feedback/edits of past year Proteopedia ProjectsTips from feedback/edits of past year Proteopedia Projects
Each section should start with the line that inserts the Jmol window: then each scene for that section will appear in that window, along side your text (<300 words).
Every jmol window should have a caption so we know what we are looking at (include the name of the molecule and pdb code) Replace 'insert caption here' with 'your caption'.
Careful with repetition of the same points in multiple sections -- instead organize the topics logically and you can have multiple people contribute to a section if you want.
Feel free to work together on sections and add people to the credits if that helps to make a coherent and organized story.
Follow the correct format for references, including citations in text -- see instructions and link above.
Make green scenes to illustrate your points, and weave your scenes into the text.
Use colored text to help the reader easily see your points in the scene and to keep your text concise. For example "This view shows the 2 alpha helices packed against the 4-stranded antiparallel beta sheet." The word view would link to a scene in which the alpha helices are red and the beta sheet is blue. Go into edit mode on this page to copy the colored text section for use on your page. See also Help:Color_Keys.
Avoid a list of miscellaneous facts. Choose the most interesting points to tell us in some detail and illustrate with green scenes.
Tell your story with green scenes and minimal text!
If you are having trouble labeling individual amino acids residues, load your molecule, go to "selections," input the amino acid, residue number, and an atom only contained once in the amino acid (must typically nitrogen), and replace your selection. Then go to "labels" and chose which label you want. This should give you a single label.
ExampleExample
This is a complex between a macromolecule and its ligand (but this ligand is not a drug) that illlustrates the use of green scenes:
|
Asp Receptor Ligand-binding domain
Overall structure
The ligand binding domain of the aspartate receptor () ) is a dimer of two 4-helix bundles that is shown here with the bound.[1] In this the N and C termini are at the bottom of the structure; this is where the connections to the transmembrane helices have been truncated.
this is out
Ligand binding site
When the protein is colored according to , residues at the ligand site are the most conserved.
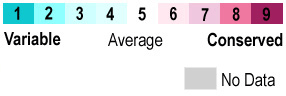 Interactions that stabilize ligand binding[2] include hydrogen bonding from Tyr149 and Gln152 backbone carbonyls and Thr154 sidechain OH to the and hydrogen bonding from the sidechain nitrogens of Arg64, Arg69, and Arg73 to the two .
Interactions that stabilize ligand binding[2] include hydrogen bonding from Tyr149 and Gln152 backbone carbonyls and Thr154 sidechain OH to the and hydrogen bonding from the sidechain nitrogens of Arg64, Arg69, and Arg73 to the two .
References
- ↑ Yeh JI, Biemann HP, Pandit J, Koshland DE, Kim SH. The three-dimensional structure of the ligand-binding domain of a wild-type bacterial chemotaxis receptor. Structural comparison to the cross-linked mutant forms and conformational changes upon ligand binding. J Biol Chem. 1993 May 5;268(13):9787-92. PMID:8486661
- ↑ Milburn MV, Prive GG, Milligan DL, Scott WG, Yeh J, Jancarik J, Koshland DE Jr, Kim SH. Three-dimensional structures of the ligand-binding domain of the bacterial aspartate receptor with and without a ligand. Science. 1991 Nov 29;254(5036):1342-7. PMID:1660187
Teams, Topics, and Links 2016Teams, Topics, and Links 2016
under construction...