Succinyl-CoA synthetase
|
Succinyl-Coa synthetase catalyzes the reversible reaction of succinyl-CoA + NDP + Pi <-> succinate + CoA + NTP (where N is either adenosine or guanosine. It can be found in Escherichia coli.
StructureStructure
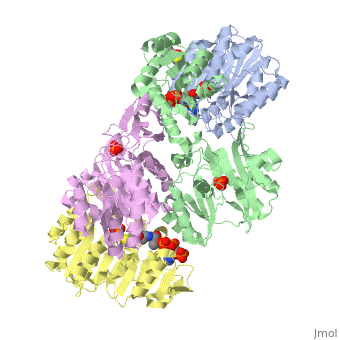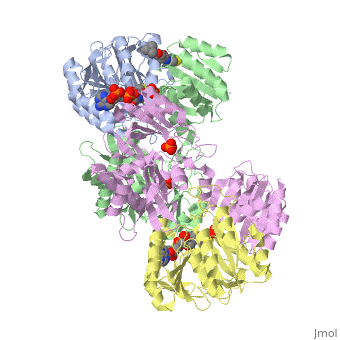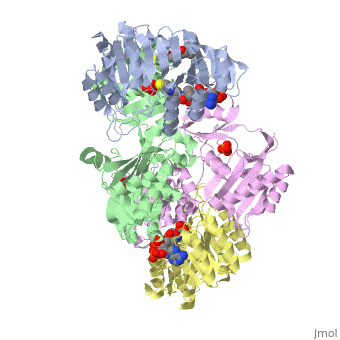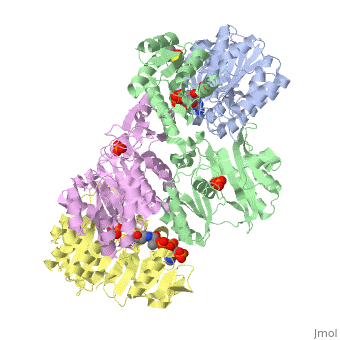
Succinyl-CoA synthetase is a tetramer with an active site on each subunit. This can be seen in the Jmol representation (the PDB code is 1CQJ). The two subunits are denoted alpha and beta. A phosphorylated histidine intermediate is responsible for the dephosphorylation of ATP and it is suspected that there is another active site on the beta subunit that is responsible for the continued catalysis of the reaction. There is a suspected nucleotide binding site on the N-terminal of the beta subunit which would imply that there are two active sites roughly 35 A apart from each other and that the HIS 246 alpha loop moves between them during catalysis. A cooperative binding catalysis mechanism has been proposed and it has been shown that binding of ATP at one catalytic site promotes catalytic activity at another catalysis site. On the alpha subunit it has been shown that GLU 208 alpha interacts with the active HIS 246 alpha residue in the phosphorylated and dephosphorylated enzyme and it is supposed that GLU 197 beta serves a similar purpose on the beta subunit.
Trypsin-BPTI complexTrypsin-BPTI complex
The trypsin backbone is shown in pink and the trypsin inhibitor, BPTI, in yellow (PDB code 2ptc). The residues [Ser195-His57-Asp102-Ser214] are shown in green, the disulfide bond between residues 14-38 is shown in yellow and the Lys 15 sidechain at the specificity site in pink.