Succinyl-CoA synthetase
|
Succinyl-Coa synthetase catalyzes the reversible reaction of succinyl-CoA + NDP + Pi <-> succinate + CoA + NTP (where N is either adenosine or guanosine. It can be found in Escherichia coli. It is the fifth step in the citric acid cycle.
StructureStructure
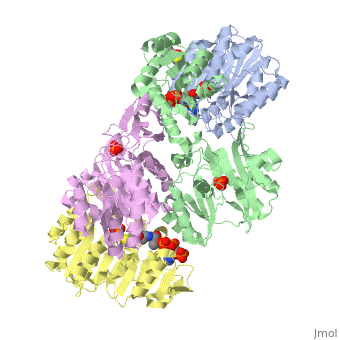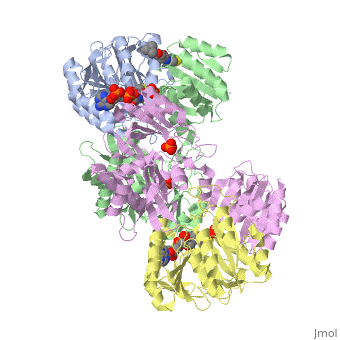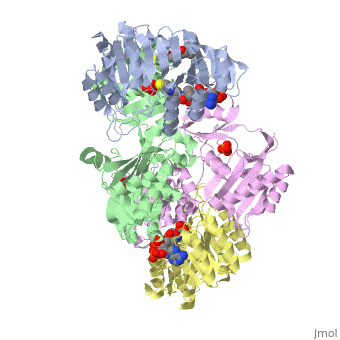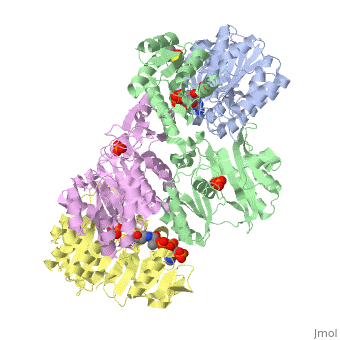
Succinyl-CoA synthetase is a tetramer with an active site on each subunit. This can be seen in the Jmol representation (the PDB code is 1CQJ). The two subunits are denoted alpha and beta. A phosphorylated histidine intermediate is responsible for the dephosphorylation of ATP and it is suspected that there is another active site on the beta subunit that is responsible for the continued catalysis of the reaction. There is a suspected nucleotide binding site on the N-terminal of the beta subunit which would imply that there are two active sites roughly 35 A apart from each other and that the HIS 246 alpha loop moves between them during catalysis.[1] On the alpha subunit it has been shown that interacts in some way with the active HIS 246 alpha residue in the phosphorylated and dephosphorylated enzyme and it is supposed that GLU 197 beta serves a similar purpose on the beta subunit.[2]
MechanismMechanism
The histidine residue involved in the (de)phosphorylation of (ATP)ADP acts through the following mechanism. File:Synthetase mechanism.jpg
A cooperative binding catalysis mechanism has been proposed and it has been shown that binding of ATP at one catalytic site promotes catalytic activity at another catalysis site.[3] It has been shown that the enzyme will bind with ATP in the presence of Mg+2 to form a complex containing 2 ATP residues as well as 2 phosphoric acid residues, after incubation this the complex converts to another one containing 4 phosphoric residues per protein. Only the second complex reacts with succinate and CoA to form the succinyl-CoA complex which then releases as many phosphoric residues as bound succinate.[4] A transfer of the phosphoric residue from the first active site is seen to be coordinate with a transfer of a phosphoric residue to the second active site suggesting again a cooperative binding catalysis. This cooperative catalysis means that the presence of ATP or ADP can be both activating and inhibiting depending on the stage of catalysis they interact with the enzyme.
After the Succinyl-CoA has been phosphorylated and subsequently dephosphorylated it is release as succinate which continues along the Krebs cycle.
Bound Form of Succinyl-CoA synthetaseBound Form of Succinyl-CoA synthetase
|
The form of succinyl-CoA synthetase shown here (PDB code 1CQI) is the bound form with two (the ADP is highlighted and the Mg is in green).[5] As seen here, the residues on both the alpha and beta subunits are present and interacting with the lone Pi group while the ADP group is bound elsewhere on the subunit.
KineticsKinetics
As stated earlier, the presence of GTP, GDP, ATP, and ADP can be either activating or inhibiting depending on the stage of catalysis it interacts with the enzyme.
RegulationRegulation
Succinyl-CoA synthetase is not a major regulator in the Krebs cycle, making it dependent on the steps prior.
Additional ResourcesAdditional Resources
For Additional Information, See Carbohydrate Metabolism
ReferencesReferences
- ↑ Joyce MA, Fraser ME, Brownie ER, James MN, Bridger WA, Wolodko WT. Probing the nucleotide-binding site of Escherichia coli succinyl-CoA synthetase. Biochemistry. 1999 Jun 1;38(22):7273-83. PMID:10353839 doi:10.1021/bi990527s
- ↑ Fraser ME, Joyce MA, Ryan DG, Wolodko WT. Two glutamate residues, Glu 208 alpha and Glu 197 beta, are crucial for phosphorylation and dephosphorylation of the active-site histidine residue in succinyl-CoA synthetase. Biochemistry. 2002 Jan 15;41(2):537-46. PMID:11781092
- ↑ Bild GS, Janson CA, Boyer PD. Subunit interaction during catalysis. ATP modulation of catalytic steps in the succinyl-CoA synthetase reaction. J Biol Chem. 1980 Sep 10;255(17):8109-15. PMID:6997289
- ↑ Mikeladze DG, Matveeva LN, Severin SE. [Reaction mechanism of succinyl CoA synthetase from pigeon thoracic muscle] Biokhimiia. 1978 Aug;43(8):1458-67. PMID:570066
- ↑ Joyce MA, Fraser ME, James MN, Bridger WA, Wolodko WT. ADP-binding site of Escherichia coli succinyl-CoA synthetase revealed by x-ray crystallography. Biochemistry. 2000 Jan 11;39(1):17-25. PMID:10625475