3a0b: Difference between revisions
No edit summary |
No edit summary |
||
| Line 10: | Line 10: | ||
</table> | </table> | ||
== Function == | == Function == | ||
[[http://www.uniprot.org/uniprot/ | [[http://www.uniprot.org/uniprot/PSBL_THEVL PSBL_THEVL]] This protein is a component of the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01317] [[http://www.uniprot.org/uniprot/PSBA_THEVL PSBA_THEVL]] D1 (PsbA) and D2 (PsbD) bind P680, the primary electron donor of photosystem II (PSII) as well as electron acceptors. PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01379] [[http://www.uniprot.org/uniprot/CY550_THEVL CY550_THEVL]] Low-potential cytochrome c that plays a role in the oxygen-evolving complex of photosystem II (PSII). Binds to PSII in the absence of other extrinsic proteins; required for binding of the PsbU protein to photosystem II. In PSII particles without oxygen-evolving activity, maximal activity is restored only by binding of cytochrome c550, PsbU and the 33 kDa PsbO protein. PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.<ref>PMID:1314738</ref> <ref>PMID:8382523</ref> [[http://www.uniprot.org/uniprot/PSBF_THEVL PSBF_THEVL]] This b-type cytochrome is tightly associated with the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00643] [[http://www.uniprot.org/uniprot/PSBJ_THEVL PSBJ_THEVL]] This protein is a component of the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01305] [[http://www.uniprot.org/uniprot/PSBT_THEVL PSBT_THEVL]] Seems to play a role in the dimerization of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00808] [[http://www.uniprot.org/uniprot/PSBE_THEVL PSBE_THEVL]] This b-type cytochrome is tightly associated with the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00642] | ||
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
Check<jmol> | Check<jmol> | ||
<jmolCheckbox> | <jmolCheckbox> | ||
<scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/a0/3a0b_consurf.spt"</scriptWhenChecked> | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/a0/3a0b_consurf.spt"</scriptWhenChecked> | ||
<scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
<text>to colour the structure by Evolutionary Conservation</text> | <text>to colour the structure by Evolutionary Conservation</text> | ||
Revision as of 10:24, 18 October 2018
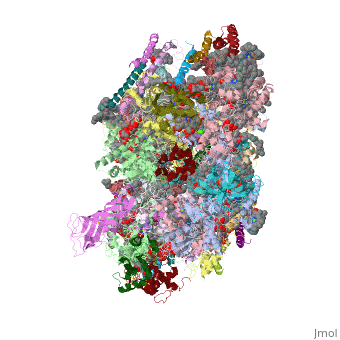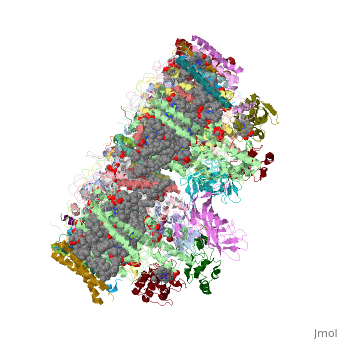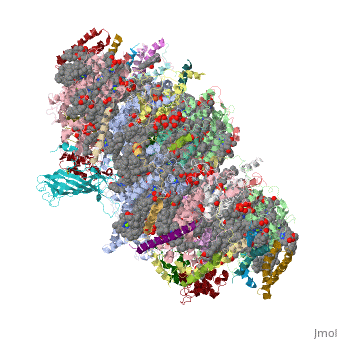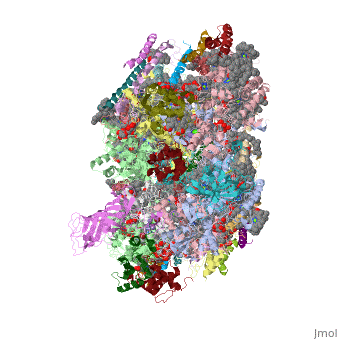
Crystal structure of Br-substituted Photosystem II complexCrystal structure of Br-substituted Photosystem II complex
Structural highlights
Function[PSBL_THEVL] This protein is a component of the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01317] [PSBA_THEVL] D1 (PsbA) and D2 (PsbD) bind P680, the primary electron donor of photosystem II (PSII) as well as electron acceptors. PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01379] [CY550_THEVL] Low-potential cytochrome c that plays a role in the oxygen-evolving complex of photosystem II (PSII). Binds to PSII in the absence of other extrinsic proteins; required for binding of the PsbU protein to photosystem II. In PSII particles without oxygen-evolving activity, maximal activity is restored only by binding of cytochrome c550, PsbU and the 33 kDa PsbO protein. PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[1] [2] [PSBF_THEVL] This b-type cytochrome is tightly associated with the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00643] [PSBJ_THEVL] This protein is a component of the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_01305] [PSBT_THEVL] Seems to play a role in the dimerization of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00808] [PSBE_THEVL] This b-type cytochrome is tightly associated with the reaction center of photosystem II (PSII). PSII is a light-driven water plastoquinone oxidoreductase, using light energy to abstract electrons from H(2)O, generating a proton gradient subsequently used for ATP formation.[HAMAP-Rule:MF_00642] Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. Publication Abstract from PubMedThe chloride ion, Cl(-), is an essential cofactor for oxygen evolution of photosystem II (PSII) and is closely associated with the Mn(4)Ca cluster. Its detailed location and function have not been identified, however. We substituted Cl(-) with a bromide ion (Br(-)) or an iodide ion (I(-)) in PSII and analyzed the crystal structures of PSII with Br(-) and I(-) substitutions. Substitution of Cl(-) with Br(-) did not inhibit oxygen evolution, whereas substitution of Cl(-) with I(-) completely inhibited oxygen evolution, indicating the efficient replacement of Cl(-) by I(-). PSII with Br(-) and I(-) substitutions were crystallized, and their structures were analyzed. The results showed that there are 2 anion-binding sites in each PSII monomer; they are located on 2 sides of the Mn(4)Ca cluster at equal distances from the metal cluster. Anion-binding site 1 is close to the main chain of D1-Glu-333, and site 2 is close to the main chain of CP43-Glu-354; these 2 residues are coordinated directly with the Mn(4)Ca cluster. In addition, site 1 is located in the entrance of a proton exit channel. These results indicate that these 2 Cl(-) anions are required to maintain the coordination structure of the Mn(4)Ca cluster as well as the proposed proton channel, thereby keeping the oxygen-evolving complex fully active. Location of chloride and its possible functions in oxygen-evolving photosystem II revealed by X-ray crystallography.,Kawakami K, Umena Y, Kamiya N, Shen JR Proc Natl Acad Sci U S A. 2009 May 26;106(21):8567-72. Epub 2009 May 11. PMID:19433803[3] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences
|
| ||||||||||||||||||||