1hyb: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
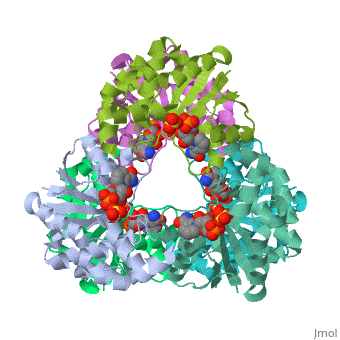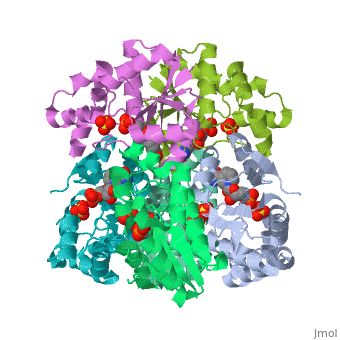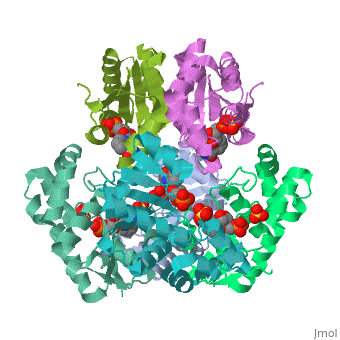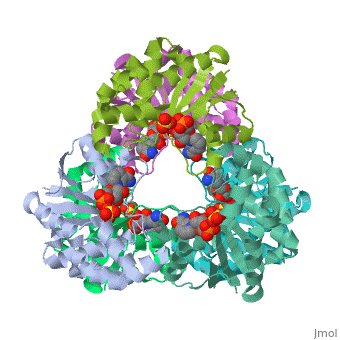
[[Image: | ==CRYSTAL STRUCTURE OF AN ACTIVE SITE MUTANT OF METHANOBACTERIUM THERMOAUTOTROPHICUM NICOTINAMIDE MONONUCLEOTIDE ADENYLYLTRANSFERASE== | ||
<StructureSection load='1hyb' size='340' side='right' caption='[[1hyb]], [[Resolution|resolution]] 2.00Å' scene=''> | |||
== Structural highlights == | |||
<table><tr><td colspan='2'>[[1hyb]] is a 1 chain structure with sequence from [http://en.wikipedia.org/wiki/Methanothermobacter_thermautotrophicus Methanothermobacter thermautotrophicus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1HYB OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1HYB FirstGlance]. <br> | |||
</td></tr><tr><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=NMN:BETA-NICOTINAMIDE+RIBOSE+MONOPHOSPHATE'>NMN</scene>, <scene name='pdbligand=SO4:SULFATE+ION'>SO4</scene><br> | |||
<tr><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat">[[1ej2|1ej2]]</td></tr> | |||
<tr><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Nicotinamide-nucleotide_adenylyltransferase Nicotinamide-nucleotide adenylyltransferase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.7.7.1 2.7.7.1] </span></td></tr> | |||
<tr><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1hyb FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1hyb OCA], [http://www.rcsb.org/pdb/explore.do?structureId=1hyb RCSB], [http://www.ebi.ac.uk/pdbsum/1hyb PDBsum]</span></td></tr> | |||
<table> | |||
== Evolutionary Conservation == | |||
[[Image:Consurf_key_small.gif|200px|right]] | |||
Check<jmol> | |||
<jmolCheckbox> | |||
<scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/hy/1hyb_consurf.spt"</scriptWhenChecked> | |||
<scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | |||
<text>to colour the structure by Evolutionary Conservation</text> | |||
</jmolCheckbox> | |||
</jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/chain_selection.php?pdb_ID=2ata ConSurf]. | |||
<div style="clear:both"></div> | |||
<div style="background-color:#fffaf0;"> | |||
== Publication Abstract from PubMed == | |||
Nicotinamide mononucleotide adenylyltransferase (NMNATase) catalyzes the linking of NMN(+) or NaMN(+) with ATP, which in all organisms is one of the common step in the synthesis of the ubiquitous coenzyme NAD(+), via both de novo and salvage biosynthetic pathways. The structure of Methanobacterium thermoautotrophicum NMNATase determined using multiwavelength anomalous dispersion phasing revealed a nucleotide-binding fold common to nucleotidyltransferase proteins. An NAD(+) molecule and a sulfate ion were bound in the active site allowing the identification of residues involved in product binding. In addition, the role of the conserved (16)HXGH(19) active site motif in catalysis was probed by mutagenic, enzymatic and crystallographic techniques, including the characterization of an NMN(+)/SO4(2-) complex of mutant H19A NMNATase. | |||
Insights into ligand binding and catalysis of a central step in NAD+ synthesis: structures of Methanobacterium thermoautotrophicum NMN adenylyltransferase complexes.,Saridakis V, Christendat D, Kimber MS, Dharamsi A, Edwards AM, Pai EF J Biol Chem. 2001 Mar 9;276(10):7225-32. Epub 2000 Nov 3. PMID:11063748<ref>PMID:11063748</ref> | |||
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |||
</div> | |||
==See Also== | ==See Also== | ||
*[[Nicotinamide mononucleotide adenylyltransferase|Nicotinamide mononucleotide adenylyltransferase]] | *[[Nicotinamide mononucleotide adenylyltransferase|Nicotinamide mononucleotide adenylyltransferase]] | ||
== References == | |||
== | <references/> | ||
< | __TOC__ | ||
</StructureSection> | |||
[[Category: Methanothermobacter thermautotrophicus]] | [[Category: Methanothermobacter thermautotrophicus]] | ||
[[Category: Nicotinamide-nucleotide adenylyltransferase]] | [[Category: Nicotinamide-nucleotide adenylyltransferase]] | ||
Revision as of 12:52, 28 September 2014
CRYSTAL STRUCTURE OF AN ACTIVE SITE MUTANT OF METHANOBACTERIUM THERMOAUTOTROPHICUM NICOTINAMIDE MONONUCLEOTIDE ADENYLYLTRANSFERASECRYSTAL STRUCTURE OF AN ACTIVE SITE MUTANT OF METHANOBACTERIUM THERMOAUTOTROPHICUM NICOTINAMIDE MONONUCLEOTIDE ADENYLYLTRANSFERASE
Structural highlights
Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. Publication Abstract from PubMedNicotinamide mononucleotide adenylyltransferase (NMNATase) catalyzes the linking of NMN(+) or NaMN(+) with ATP, which in all organisms is one of the common step in the synthesis of the ubiquitous coenzyme NAD(+), via both de novo and salvage biosynthetic pathways. The structure of Methanobacterium thermoautotrophicum NMNATase determined using multiwavelength anomalous dispersion phasing revealed a nucleotide-binding fold common to nucleotidyltransferase proteins. An NAD(+) molecule and a sulfate ion were bound in the active site allowing the identification of residues involved in product binding. In addition, the role of the conserved (16)HXGH(19) active site motif in catalysis was probed by mutagenic, enzymatic and crystallographic techniques, including the characterization of an NMN(+)/SO4(2-) complex of mutant H19A NMNATase. Insights into ligand binding and catalysis of a central step in NAD+ synthesis: structures of Methanobacterium thermoautotrophicum NMN adenylyltransferase complexes.,Saridakis V, Christendat D, Kimber MS, Dharamsi A, Edwards AM, Pai EF J Biol Chem. 2001 Mar 9;276(10):7225-32. Epub 2000 Nov 3. PMID:11063748[1] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences
|
| ||||||||||||||||||||