5dfr: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
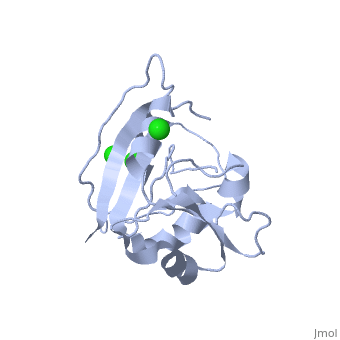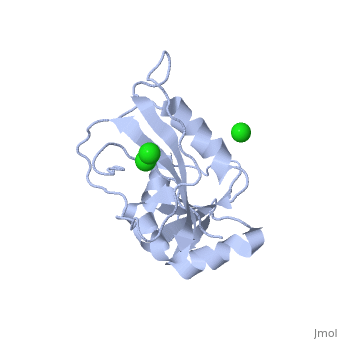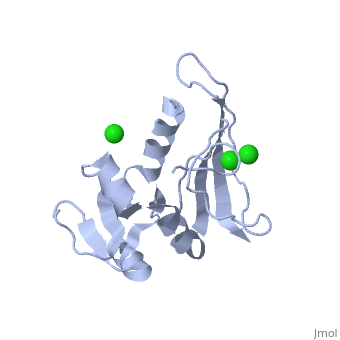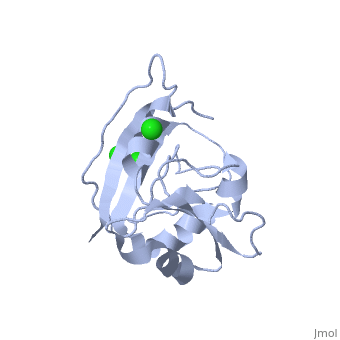
==CRYSTAL STRUCTURE OF UNLIGANDED ESCHERICHIA COLI DIHYDROFOLATE REDUCTASE. LIGAND-INDUCED CONFORMATIONAL CHANGES AND COOPERATIVITY IN BINDING== | ==CRYSTAL STRUCTURE OF UNLIGANDED ESCHERICHIA COLI DIHYDROFOLATE REDUCTASE. LIGAND-INDUCED CONFORMATIONAL CHANGES AND COOPERATIVITY IN BINDING== | ||
<StructureSection load='5dfr' size='340' side='right' caption='[[5dfr]], [[Resolution|resolution]] 2.30Å' scene=''> | <StructureSection load='5dfr' size='340' side='right' caption='[[5dfr]], [[Resolution|resolution]] 2.30Å' scene=''> | ||
| Line 5: | Line 6: | ||
</td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene></td></tr> | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene></td></tr> | ||
<tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Dihydrofolate_reductase Dihydrofolate reductase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=1.5.1.3 1.5.1.3] </span></td></tr> | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Dihydrofolate_reductase Dihydrofolate reductase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=1.5.1.3 1.5.1.3] </span></td></tr> | ||
<tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=5dfr FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=5dfr OCA], [http://pdbe.org/5dfr PDBe], [http://www.rcsb.org/pdb/explore.do?structureId=5dfr RCSB], [http://www.ebi.ac.uk/pdbsum/5dfr PDBsum]</span></td></tr> | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=5dfr FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=5dfr OCA], [http://pdbe.org/5dfr PDBe], [http://www.rcsb.org/pdb/explore.do?structureId=5dfr RCSB], [http://www.ebi.ac.uk/pdbsum/5dfr PDBsum], [http://prosat.h-its.org/prosat/prosatexe?pdbcode=5dfr ProSAT]</span></td></tr> | ||
</table> | </table> | ||
== Function == | == Function == | ||
| Line 30: | Line 31: | ||
==See Also== | ==See Also== | ||
*[[Molecular Playground/DHFR|Molecular Playground/DHFR]] | *[[Molecular Playground/DHFR|Molecular Playground/DHFR]] | ||
== References == | == References == | ||
Revision as of 10:22, 29 November 2017
CRYSTAL STRUCTURE OF UNLIGANDED ESCHERICHIA COLI DIHYDROFOLATE REDUCTASE. LIGAND-INDUCED CONFORMATIONAL CHANGES AND COOPERATIVITY IN BINDINGCRYSTAL STRUCTURE OF UNLIGANDED ESCHERICHIA COLI DIHYDROFOLATE REDUCTASE. LIGAND-INDUCED CONFORMATIONAL CHANGES AND COOPERATIVITY IN BINDING
Structural highlights
Function[DYR_ECOLI] Key enzyme in folate metabolism. Catalyzes an essential reaction for de novo glycine and purine synthesis, and for DNA precursor synthesis. Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. Publication Abstract from PubMedThe crystal structure of unliganded dihydrofolate reductase (DHFR) from Escherichia coli has been solved and refined to an R factor of 19% at 2.3-A resolution in a crystal form that is nonisomorphous with each of the previously reported E. coli DHFR crystal structures [Bolin, J. T., Filman, D. J., Matthews, D. A., Hamlin, B. C., & Kraut, J. (1982) J. Biol. Chem. 257, 13650-13662; Bystroff, C., Oatley, S. J., & Kraut, J. (1990) Biochemistry 29, 3263-3277]. Significant conformational changes occur between the apoenzyme and each of the complexes: the NADP+ holoenzyme, the folate-NADP+ ternary complex, and the methotrexate (MTX) binary complex. The changes are small, with the largest about 3 A and most of them less than 1 A. For simplicity a two-domain description is adopted in which one domain contains the NADP+ 2'-phosphate binding site and the binding sites for the rest of the coenzyme and for the substrate lie between the two domains. Binding of either NADP+ or MTX induces a closing of the PABG-binding cleft and realignment of alpha-helices C and F which bind the pyrophosphate of the coenzyme. Formation of the ternary complex from the holoenzyme does not involve further relative domain shifts but does involve a shift of alpha-helix B and a floppy loop (the Met-20 loop) that precedes alpha B. These observations suggest a mechanism for cooperativity in binding between substrate and coenzyme wherein the greatest degree of cooperativity is expressed in the transition-state complex. We explore the idea that the MTX binary complex in some ways resembles the transition-state complex. Crystal structure of unliganded Escherichia coli dihydrofolate reductase. Ligand-induced conformational changes and cooperativity in binding.,Bystroff C, Kraut J Biochemistry. 1991 Feb 26;30(8):2227-39. PMID:1998681[1] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. See AlsoReferences |
| ||||||||||||||||||