1st6: Difference between revisions
Jump to navigation
Jump to search
No edit summary |
No edit summary |
||
| (3 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
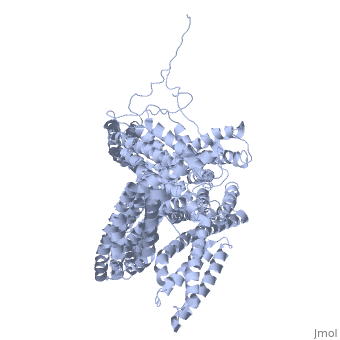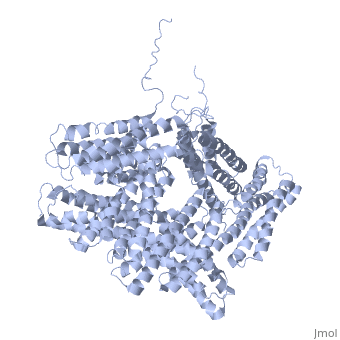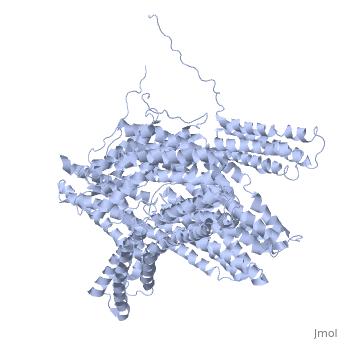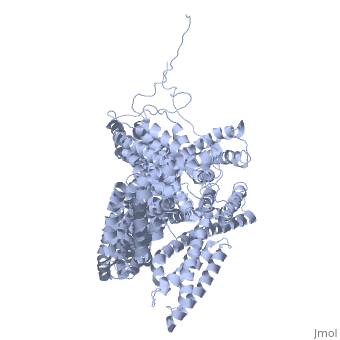
==Crystal structure of a cytoskeletal protein== | ==Crystal structure of a cytoskeletal protein== | ||
<StructureSection load='1st6' size='340' side='right' caption='[[1st6]], [[Resolution|resolution]] 3.10Å' scene=''> | <StructureSection load='1st6' size='340' side='right'caption='[[1st6]], [[Resolution|resolution]] 3.10Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
<table><tr><td colspan='2'>[[1st6]] is a 1 chain structure with sequence from [ | <table><tr><td colspan='2'>[[1st6]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Gallus_gallus Gallus gallus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1ST6 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1ST6 FirstGlance]. <br> | ||
</td></tr><tr id=' | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 3.1Å</td></tr> | ||
<tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1st6 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1st6 OCA], [https://pdbe.org/1st6 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1st6 RCSB], [https://www.ebi.ac.uk/pdbsum/1st6 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1st6 ProSAT]</span></td></tr> | ||
</table> | </table> | ||
== Function == | == Function == | ||
[ | [https://www.uniprot.org/uniprot/VINC_CHICK VINC_CHICK] Actin filament (F-actin)-binding protein involved in cell-matrix adhesion and cell-cell adhesion. Regulates cell-surface E-cadherin expression and potentiates mechanosensing by the E-cadherin complex. May also play important roles in cell morphology and locomotion.<ref>PMID:15229287</ref> <ref>PMID:20584916</ref> <ref>PMID:20086044</ref> | ||
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
Check<jmol> | Check<jmol> | ||
<jmolCheckbox> | <jmolCheckbox> | ||
<scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/st/1st6_consurf.spt"</scriptWhenChecked> | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/st/1st6_consurf.spt"</scriptWhenChecked> | ||
<scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
<text>to colour the structure by Evolutionary Conservation</text> | <text>to colour the structure by Evolutionary Conservation</text> | ||
</jmolCheckbox> | </jmolCheckbox> | ||
</jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/ | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1st6 ConSurf]. | ||
<div style="clear:both"></div> | <div style="clear:both"></div> | ||
==See Also== | ==See Also== | ||
| Line 34: | Line 27: | ||
</StructureSection> | </StructureSection> | ||
[[Category: Gallus gallus]] | [[Category: Gallus gallus]] | ||
[[Category: | [[Category: Large Structures]] | ||
[[Category: | [[Category: Bakolitsa C]] | ||
[[Category: | [[Category: Liddington RC]] | ||
Latest revision as of 11:33, 14 February 2024
Crystal structure of a cytoskeletal proteinCrystal structure of a cytoskeletal protein
Structural highlights
FunctionVINC_CHICK Actin filament (F-actin)-binding protein involved in cell-matrix adhesion and cell-cell adhesion. Regulates cell-surface E-cadherin expression and potentiates mechanosensing by the E-cadherin complex. May also play important roles in cell morphology and locomotion.[1] [2] [3] Evolutionary Conservation Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf. See AlsoReferences
|
| ||||||||||||||||